新生儿鉴定的生物识别指纹工具
加州大学圣地亚哥分校研究人员的研究人员创建了一种新的生物识别技术,可以捕获婴儿和儿童的指纹,并只用手指浪潮来识别它们。“我们认为我们已经解决了发达国家和发展中国家的婴儿识别问题,”加州大学圣地亚哥分校医学院医学助理教授MD,医学博士Eliah Aronoff-Spencer说。“这项新技术允许快速,准确的指纹打印,可以消除对纸张识别的需求,并改善数百万的医疗保健和安全性。”
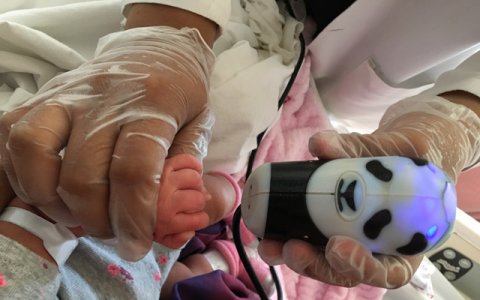
在全球范围内,卫生保健提供的婴儿和童年鉴定是需要的,尤其是在偏远或资源有限的地区,以及支持救灾,人口贩运,移民和难民解决方案的努力。
Spencer said other technologists have unsuccessfully attempted to extrapolate adult technologies to fingerprinting children. His solution was to apply a human-centered design and develop the technology from the ground up with infants, caregivers and stakeholders in mind. “Accurate identification of a child to enable timely vaccinations can improve care, reduce disease burden and save lives,” said Spencer. This technology could be usefulness in tracking vaccinations and aid or prevent infectious disease outbreaks. A person’s identity could now be secured at birth, potentially protecting from identity fraud many years in the future. “Imagine the ability to assist refugees displaced by war or natural disasters to establish their identity so they can access needed food, aid and care. While a difficult subject, this technology may have applications to help prevent human trafficking and other causes of separation of children from their families.”
Non-contact optical scanning technology
Spencer said the technology was conceived to accommodate the size, movements and behaviors of an infant. “Not only did we take into account the child’s physiology and reflexes, but also what would be culturally acceptable in different countries. For example, in some areas, facial photography is shunned, but photography of hands is acceptable.”
该设备称为离子,是一种非接触式光学扫描技术,可以对手指和手掌进行成像。印刷品被存储为可以在平台上安全共享的加密模板。当前的设备坚固,便携式,适合用户的手,并与笔记本电脑和移动平台一起使用。在开发该技术以识别婴儿和幼儿时,它与成年人的准确性高,使其成为所有年龄段的第一个生物识别平台。正在开发的增强功能包括还可以测量健康生物识别技术和其他临床数据,例如温度,脉搏,呼吸和氧气。
Institution Review Board-approved clinical trials are currently underway at UC San Diego and with collaborators in Mexico. Preliminary results show that the device delivers greater than 99 percent accuracy on re-identification after registration as early as two days after birth, with 90 percent accuracy for registration on the first day of birth.
“The next stage is to take the device into the field in Africa and South Asia and broaden the populations we evaluate,” said Spencer. “While the device is not yet commercially available, we hope to have it ready for market within 12 months. We want to continue to validate the platform, work through workflow, security and ethical issues, and, with funding, make the technology available on a staged basis to non-governmental organizations and government programs at local and national levels.”